This can be considered a companion piece to one I wrote previously, titled ‘How psychiatric drugs create a vicious cycle of effects, more drugs and diagnoses’. Here, I discuss how the medicalisation of ‘depression’ in the public’s awareness could lead a young person experiencing passing emotional difficulties to be started on ‘antidepressant medication’. Giving rise to a train of adverse consequences for them. I explore some of the harmful consequences that ‘selective serotonin reuptake inhibitors’ (SSRIs) can have and I also discuss the marketing and ‘evidence-based medicine’ that served to justify their use.
“What I know about depression is that, it’s an illness. They can cure it.”
By the late 1990s this was an increasingly common view among people, including the young and impressionable. During the nineties in Britain a public awareness campaign called ‘Defeat Depression’ had been launched. It ran between 1991 and 1996. Among its key objectives were to popularise this notion, and to shift negative public perceptions about taking antidepressant drugs. The addictive nature of benzodiazepines was by then well known. So people were sceptical about drug treatments for emotional difficulties.
Aside from fun runs for psychiatrists, there were newspaper and magazine articles, radio and television programmes. Professional educational materials including films were disseminated to General Practices. They encouraged prescribing the new SSRI class of drugs for milder cases of depression. These had began to be widely used after Prozac was first launched in 1988.
By the end of the Defeat Depression campaign there had been a roughly four-fold increase in the prescribing of SSRIs by GPs. A generation of psychiatrists were educated to prescribe the drugs routinely. The campaign was generously funded by SmithKline Beecham (now GlaxoSmithKline). ‘Defeat Depression’ can now be seen as a clear example of a public health education campaign being driven by drug company marketing.
The messaging filtered through into the public consciousness. In a sense, it was reassuring, suggesting that the depressed person shouldn’t feel any burden of responsibility. A ‘chemical imbalance’ in the brain isn’t a personal failing. But the notion that depression was a simple and easily resolvable neurochemical deficiency was, even at best, misleading. It would though become ‘common knowledge’, with nearly 9 out of 10 people coming to believe it.

It was this background of changing public consciousness in the late nineties that could lead a young university student, concerned by her twenty-year-old partner’s lack of motivation and apprehensiveness, to decide to do what seemed the responsible thing. Having picked up on the new official wisdom, she urged and accompanied him down to the medical practice.
After listening to her for a few minutes, the GP concluded, “Well, it does sound like he has depression.” On this basis, and without having seen him before, the doctor prescribed him the drug paroxetine. Branded as ‘Seroxat’ or as ‘Paxil’ in the US, SmithKline Beecham’s SSRI product, launched in 1991, had by then become a popular option for doctors to prescribe.
There were notable changes from the same day as he took the first pill. His pupils became enlarged. Then there could be digestive symptoms, like difficulty getting food down in the mornings. Other changes were noticed with sexual function. His partner also noticed that his body smell had changed.
His behaviour also seems to have been affected subsequently. He could be indifferent to the concerns of his cohabiting partner. He might become a bit volatile or dramatic, especially with alcohol. He didn’t show up to exams. Another time he fixed on the idea of suicide and tried taking an overdose. Within two years, in different circumstances, he (the humble author) would be hospitalised for ‘psychosis’, not long after he had restarted taking an ‘antidepressant’.
Unbeknownst to prescribers at the time, let alone young university students, the promotion of SSRIs as safe and effective was not all that it seemed to be.
Latterly, researchers have recognised that taking SSRIs can result in these kind of issues, which they term ‘SSRI-induced activation syndrome’. Said to manifest in symptoms of ‘excessive emotional arousal or behavioural activation’. To take one example, in a trial of an SSRI on youths aged 9 to 20 more than half of the participants discontinued the drugs after experiencing adverse reactions, such as irritability, aggression, impulsivity or hyperactivity.
Many now acknowledge that the drugs cause what is termed ‘emotional blunting’ or ‘indifference’. Trials appear to show that being on SSRIs can make people less responsive to feedback about their actions. I would say of that period as a 20 year-old student, that I could be less conscientious, with sensitivity about how my actions would affect people being overridden.
One of the trial studies from the nineties that made the case for paroxetine has since become notorious. A re-examination of the trial data showed it to be fraudulent. As the website study329.org documents:
…GlaxoSmithKline (GSK) marketed Paxil (paroxetine) as safe and effective for children and adolescents when company executives were aware that a number of studies (all but Study 329 unpublished) had shown that the drug was no better than placebo…
After their studies failed to show any benefit, a 1998 internal memo acknowledged that “it would be commercially unacceptable to include a statement that efficacy had not been demonstrated”. Regulatory concerns were not to stand in the way of the product’s marketing. Especially, when competitors Lilly and Pfizer were ahead in bringing their own SSRIs to the youth market.
Worse still, GSK employees suppressed trial data indicating that the drug “…caused thought disturbance and suicidal behaviour in youngsters”. As the psychiatrist David Healy has documented, various slights of hand were used when preparing the trial data for publication. For example, episodes of ‘suicidal ideation’ (feeling suicidal) were vaguely classified as ‘emotional lability’. Whilst a danger signal in the data for younger people was diluted by lumping different age groups and diagnoses together.
The drugs were already being prescribed off-label to youngsters. Ben Goldacre describes in his book ‘Bad Pharma’ — which documents systematic failings within ‘evidence-based medicine’ – that due to a loophole in the regulations for off-label uses, GSK could get away with not reporting the adverse safety data to the regulator.
Only in 2004 did regulators in Canada, the US and UK establish ‘black box’ warnings on the risk of suicidality, violence, aggression, mania and other behavioural changes in children and adolescents. Obviously, such risks don’t just vanish on a person’s eighteenth birthday, as that guidance might seem to imply. Three years later the warnings were extended to under 25s. They should have been in place when the drugs first came to market thirteen years earlier.
In 2012 GSK was fined $3bn for suppressing paroxetine’s safety data and other instances of promoting drugs misleadingly. It remains the largest fine ever on a pharma company. However, as a Guardian article, titled ‘Pharma overtakes arms industry to top the league of misbehaviour’ explained this amount was substantially smaller than the profits from the drugs concerned. The following year in the US prescribing of Paxil continued to rise unabated.
Dr Peter Breggin has argued that when exposed to drugs that affect neurotransmitters, compensatory changes occur in the brain and nervous system, as they seek to maintain stasis. For example, an uptick in available serotonin will lead to a reduction in the number of receptors for it. Whilst cells that carry out the serotonin reuptake function will proliferate — a type of abnormal brain growth. Such changes will persist long after the drug has ‘washed out’ of the system. Their long term effects on people remain poorly understood.
Another major concern with ‘SSRI induced activation syndrome’ is its link with manic type symptoms. It is now well known that SSRIs can induce mania or overstimulation in susceptible persons. In an early SSRI trial, 17% of subjects diagnosed as ‘unipolar depressed’ developed manic symptoms. Peter Breggin has suggested that increasing rates of ‘bipolar’ diagnosis are, at least partly, the consequence of the increasing use of SSRIs.
Some real-world evidence does seem to support SSRI prescriptions leading to ‘bipolar’ diagnoses. The relation seems to be clearest in young people. In the US, as this 2007 article notes, “bipolar disorder was one of the least-frequent psychiatric diagnoses recorded for child inpatients in 1996, but the most common in 2004.”
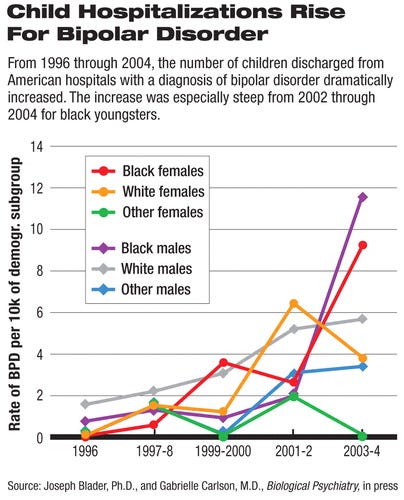
In the UK, data for 1999–2017 shows that rising rates of adolescent ‘emotional disorders’ coincided with a doubling of SSRI use in the age group. Within that time frame the average age for bipolar diagnoses fell from a person’s early 30s to their early 20s. Increasing rates of suicide and self-harm among adolescents have also been recorded since the mid-nineties.
In an example of the obtuse logic of psychiatry, it has been suggested that SSRIs can ‘reveal’ a person’s ‘latent bipolar depression’. Such a revelation will likely lead to the prescription of ‘mood stabiliser’ drugs.
The elevated risks of mania can also extend to ‘psychosis’. If a person taking SSRIs is exposed to other drugs that elevate serotonin activity, these and other serious risks will be exacerbated.
In psychiatric assessments, a ‘mania’ can be ascribed as having ‘psychotic features’, or a ‘psychosis’ as having ‘manic features’. Shifts in the criteria can have serious consequences. A patient being assessed as ‘psychotic’, where susceptibility to antidepressant-induced mania was a factor, could lead to a diagnosis like ‘schizoaffective disorder’. Perhaps resulting in the unnecessary prescribing of both a ‘mood stabiliser’ and an ‘antipsychotic’, exposing them to greater toxicity.
Potentially many other factors may be relevant in an explanation of the causes of such episodes. However, it does seem that not only do SSRIs raise the chances, but they may also make what would otherwise be a short-lived experience into a more persistent and disruptive episode.
A Google search result page informs us that the answer to why rates of bipolar diagnosis have increased is “better mental health awareness”. More people are seeking assistance, so more cases are being recognised. Many would see this ‘greater recognition’ as really representing a culture of overdiagnosis, the result of ‘disease mongering’, or a ‘psychiatrisation’ of the common range of human experience.
Professor David Healy suggests that one pathway for ever-increasing numbers receiving ‘psychiatric treatment’ is screening questionnaires and rating scales, like the widely used PHQ-9 and GAD-7 questionnaires. Such screening may lead services to ‘discover’ that their patients have ‘unrecognised bipolar disorder’, and thus require ‘mood stabilisers’. Even though they weren’t presenting to services with such symptoms prior to filling out the questionnaires.
So what then of the serotonin theory of depression? A recent umbrella review of relevant papers by Joanna Moncrieff et al, which sparked a lot of media controversy, found no evidence supporting the Serotonin theory of depression across a wide range of measures.
More simply, as Ben Goldacre has pointed out, the drug Tianeptine which enhances serotonin reuptake — the opposite action of an SSRI — is regarded as being a good antidepressant. So, a drug that lowers available serotonin is said to function well as an ‘antidepressant’, even when the theory says depression is caused by lowered serotonin.
Study 329 was far from being the only example of selective publishing of SSRI trials by drug companies, as Erick Turner has documented. Below is a slide showing the overall results of drug company SSRI trials, which the US Food and Drug Administration had access to. The total positive indication is only negligibly better than what would be expected on the basis of pure chance.
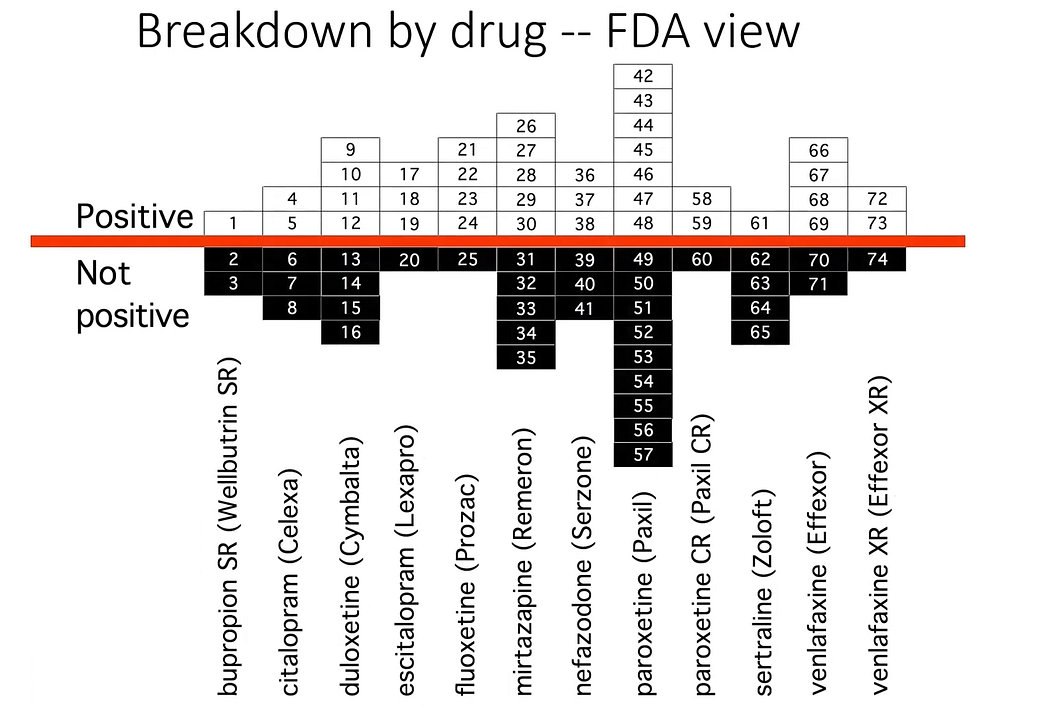
Disturbingly, only three of the reports for non-positive trials were published. Moreover, many of the non-positive trials were presented as positive. One way for a drug company to achieve this is through ‘outcomes switching’. If the primary outcomes of the trial are not positive, they look for secondary, minor outcome measures that have a positive indication, and they then present these measures as the main focus of the trial. The regulations are often lax and with enough loopholes to allow such a disingenuous presentation of data to remain within their letter.
When the SSRI trials produced disappointing results the companies had to innovate to market them. David Healy has written that:
When concerns emerged about tranquilliser dependence in the early 1980s, an attempt was made to supplant benzodiazepines with a serotonergic drug, buspirone, marketed as a non-dependence-producing anxiolytic. This flopped…Instead, drug companies marketed SSRIs for depression…and sold the idea that depression was the deeper illness behind the superficial manifestations of anxiety.
Later GSK would catch up with lost ground on its competitors by promoting paroxetine as an anti-anxiety treatment. This would involve inculcating in the public some felt awareness of ‘anxiety disorders’. Driving a message that they are common, serious, but thankfully, treatable. The marketing strategies employed by the pharma concerns were described in this article in the Guardian over twenty years ago. “First, you market the disease… then you push the pills to treat it”.
Typically, a corporate-sponsored “disease awareness” campaign focuses on a mild psychiatric condition with a large pool of potential sufferers…Prominent doctors are enlisted to publicly affirm the malady’s ubiquity, then public-relations firms launch campaigns to promote the new disease. Finally, patient groups are recruited to serve as the “public face” for the condition…The strategy has enabled the pharmaceutical industry to squeeze millions in additional revenue from the blockbuster drugs known as selective serotonin reuptake inhibitors…
SSRIs are now prescribed for ‘conditions’ related to anxiety, obsessive compulsions, panic attacks, phobias, bulimia and sometimes also premenstrual syndrome, fibromyalgia, irritable bowel syndrome and premature ejaculation.
They continue to be prescribed to young people in increasing numbers, even when top medical journals acknowledge that “…the effects of SSRIs in adolescence, a time when there are substantial changes in neural, cognitive, and social functioning, are not well understood.”
It is now clear that the drugs were promoted in spite of the companies knowing the adverse consequences they could have for a significant proportion of patients. When these harmful and escalating effects played out in society at large, healthcare services would often attribute them to ‘better recognition’ of pre-existing conditions. Ad hoc diagnoses like ‘latent’ or ‘unrecognised bipolar disorder’ entered medical texts. As I’ve argued previously, all this represents a kind of institutional dysfunction, aptly characterised as a “self-licking ice-cream cone”.
Regulatory and professional bodies whose professed first duty is, “Do no harm”, must account for allowing this to happen.

A 2004 BBC Panorama programme on Seroxat and the failure of the British regulatory agency MHRA. The programme attributes repeated oversights of the adverse effects in youths to regulators not analysing the raw trial data. They simply relied on trial summaries provided by GSK employees. https://www.youtube.com/watch?v=qknM3j1KVP0